One issue that has been discussed lately is the potential value in boosting your COVID-19 vaccination with a different vaccine from the original one you received. There is some rationale for doing this as you would potentially present an overlapping set of antigens to your immune system, and therefore, might create a wider and more protective response.
There are studies currently underway; but most have not been published. However, because many of us have been considering getting a booster in the near future, I thought it might be useful to look at the single paper that has been made public on the issue of cross-boosting.
The one paper that has been recently released has not yet been peer-reviewed and is only available as a pre-print. This means that the data has not be scrubbed by independent reviewers yet and may need to be modified before publication.
This paper comes out of Baylor U. and is, therefore, considered to be pretty reliable.
The study looked at people who had been vaccinated at least 12 weeks prior to enrollment in the study and who had not been previously infected with COVID-19.
The total number of people studied were 458 (which is considered a small sample).
They were divided into 9 groups. People who had been immunized with one of the 3 vaccines currently used were then boosted with one of each of the 3 vaccines, yielding the 9 groups.
The researchers then looked at potential “adverse events” (reactions to the booster vaccine), and the antibody levels at the point of boosting as well as on days 15 and 29 after boost.
With respect to the reactions to the vaccines, each of the 9 groups showed the same side effects as original vaccines; there was no advantage or disadvantage to any of the 9 groups when considering reactions to the boost.
The authors evaluated the stimulation of “neutralizing” antibodies (those antibodies that will prevent virus from infecting cells as measured in tissue cultures in the laboratory); their results are shown in the table below.
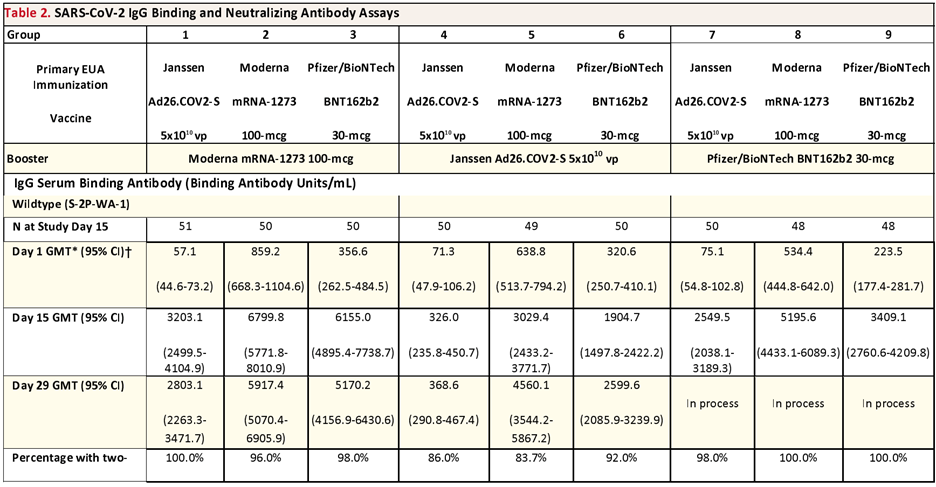
The second image is my interpretation of these results:
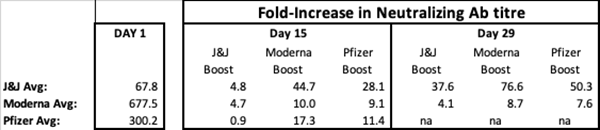
So, what do the results show?
If you were immunized with the J&J vaccine, your neutralizing antibodies were at a lower level at Day 1 than the other 2 vaccines.
Average Initial levels of neutralizing anitibodies:
J&J 67.8
Moderna 677.5
Pfizer 300.2
You should not make a conclusion from this that J&J is less or more protective than the other 2 vaccines. It is really important to always keep in mind that NO vaccine, EVER, has been designed to create a “force field” around an individual to prevent infection; they were ALWAYS designed to prevent severe disease and lower transmission. ALL three of these vaccines have been spectacularly effective in preventing severe disease, hospitalization and death as well as reducing transmissible virus shedding by factors of 1,000.
Now, looking at the data from day 15 and day 29 you can see that with Moderna, the effectiveness of boosting was seen at day 15, with little additional rise in Ab titre at 29 days. It also seems that having been originally immunized with the Moderna vaccine, that there is little advantage of boosting with any one of the three vaccines. (I don’t think that the results varying from an increased titre of between 4.1-fold and 10-fold is significant.)
Results from Pfizer at 29 days are not yet available, but because both use the same technology of mRNA vaccination, it would be expected that you would see the same results as with Moderna, i.e., that the effect would not change significantly from 15 days to 29 days. However, the implication of these results is that if you were vaccinated with Pfizer initially, there MAY be a value to cross boosting with either the Pfizer or Moderna vaccines rather than the J&J. The subject set was too low to really make a good conclusion; but the results are suggestive enough that I, personally would boost with either the Pfizer or the Moderna (whichever is most easily available) had I been originally immunized with the Pfizer vaccine.
With J&J, it appears that the effectiveness of that vaccine is most likely increased by a second shot. The lag in effectiveness may be due to the need for a second vaccination, much like the other two vectors. Again, it is important to remember that J&J never actually said that this would be a one-dose vaccine, but rather that they ONLY submitted for FDA approval after data were collected after a single dose. The FDA can ONLY RULE on the data they see, they cannot make decisions based on speculation. That is also the reason that Moderna required a second dose at 4 weeks and Pfizer at 3 weeks. It is NOT because the two companies studied second doses at 1, 2, 3, 4, 5 and 6 weeks and then concluded that a particular waiting period was appropriate, but rather that they ONLY submitted data when second doses were given at a specific date after the first dose and, therefore, it was only possible for the FDA and the CDC to approve that vaccination timing.
Also, the J&J vaccine mechanism is different from Moderna and Pfizer. J&J does not use mRNA, instead it uses DNA inserted into the genome of an adenovirus and it may be that the cellular processes needed to display the antigen (the spike protein) on the cell surface to stimulate an immune response, takes a little longer.
Nevertheless, if you received the J&J vaccine first, it appears that at day 29 all three vaccines give similar boosts, so there appears that with the J&J vaccine, any one of the three vaccines would be equally effective as a booster.
